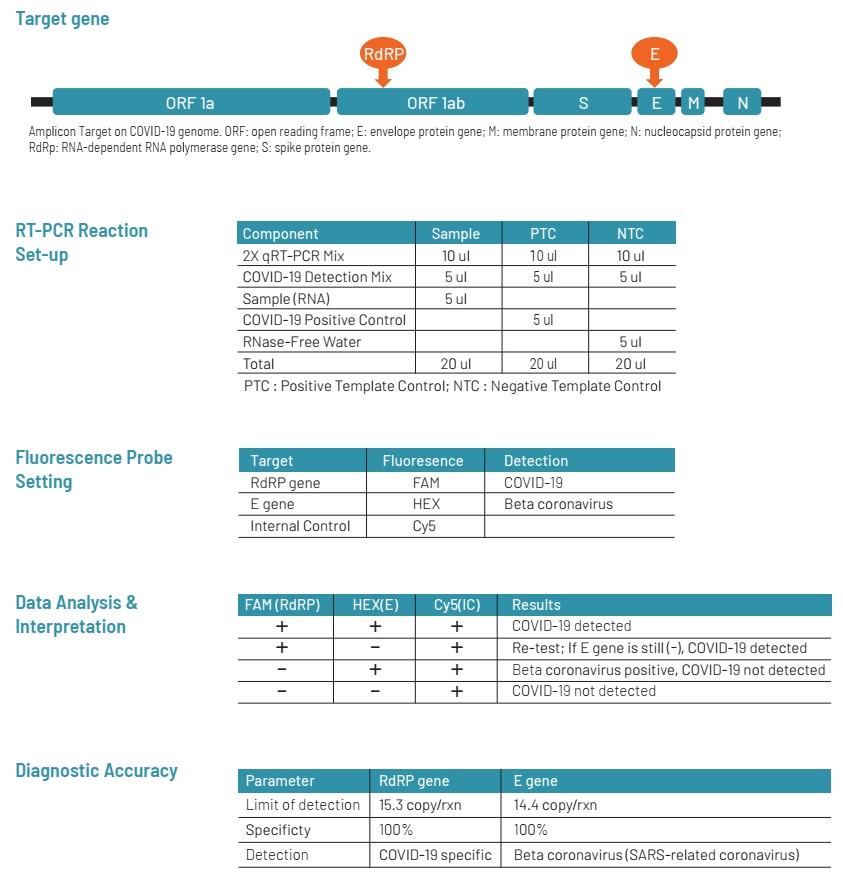
WizDx™ COVID-19 Real-time PCR Kit is intended to be used to achieve qualitative detection of 2019-Novel Coronavirus (COVID-19) viral RNA extracted from nasopharyngeal swabs, oropharyngeal swabs, and sputum from patients.
MFDS License No. : IVD-20-330
Download
Download