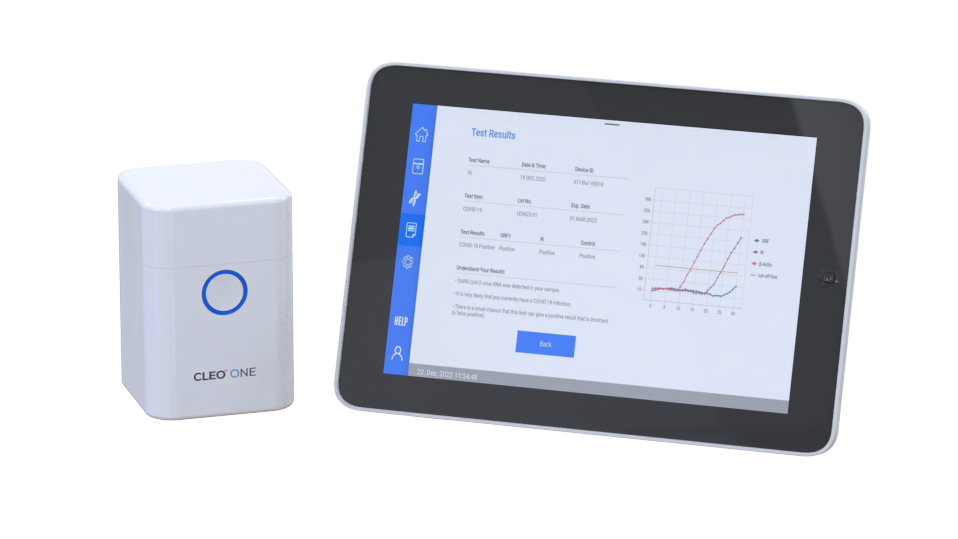

The CLEO™ ONE system is intended to perform for nucleic acid amplification of samples.
CLEO™ ONE system is intended for in vitro diagnostic use only.
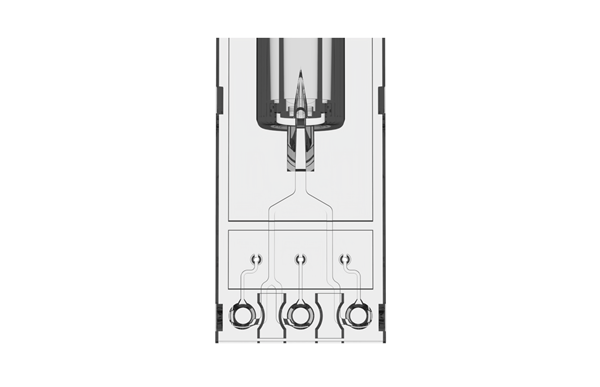
The CLEO™ ONE system is a compact molecular amplification instrument with fluorescence detection.
MFDS License No. : IVD-23-4812
For more information, please visit www.cleo1.net
Download
Download
| Model | CLEO™ ONE | |
| Test capacity | 1 test | |
| Detection channel | 1 channel (FAM) | |
| Reaction chamber | 3 well | |
| Connection | Blooth 5.0 | |
| Test method | Fluorescence-Isothermal amplification | |
| Test sample | Nasal swab | |
| Operating system | Android OS, iOS | |
| Power supply | 100 - 240V, 50/60Hz, 2A | |
| Dimensions (LxWxH) | 72×72×104 mm (2.8″x2.8″x4.1″) | |
| Weight | 165g (5.8 oz) |